|
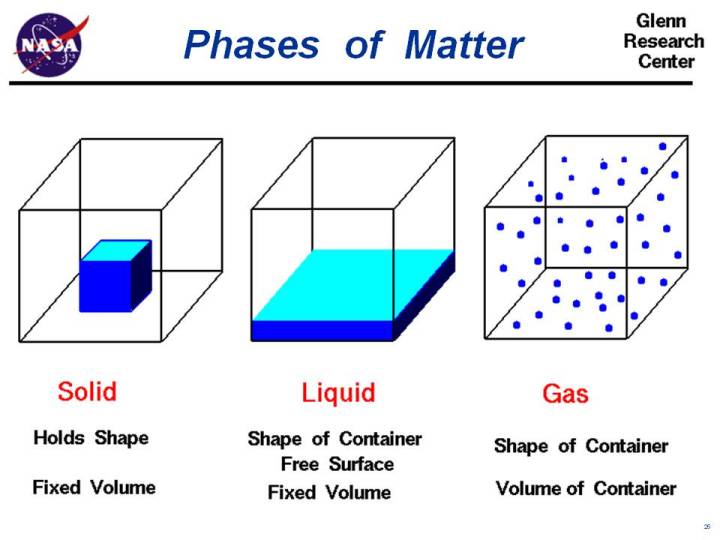
We were taught in school about what matter is. Look around you. The whole thing you see is matter. Can you recall the definition? Matter is anything that has mass and volume (occupies space). From the definition of matter, let us define some terms in order for you to clearly understand what matter is. Mass is the amount of matter in an object. Keep in mind that weight is the force of gravity pulling on the object. An object's weight is dependent upon mass and gravity pulling on it. All matter takes up space. This means it has volume. For the moment, let us side-step the meaning of matter and focus on the chemist’s view: matter is what chemical substances are composed of. But what do we connote by chemical substances? How do we sort out our view of matter and its properties? These will be the area under discussion of this lesson. Phases of MatterNot all matter is identical. Look at the different kinds of matter in your surroundings. The chairs, notebooks, tables, the air that we breathe and the water that we drink are all different forms or states of matter. Scientists call these form of matter its phase. There are four phases of matter and these are: solids, liquids,and gases – phases of matter commonly found on Earth. Plasma is the fourth phase of matter. 1. Solid
2. Liquid
3. Gas
4. PlasmaFound in stars and common in the universe. On Earth, plasmas usually do not take place naturally except in parts of flames and in lightning bolts. Properties of MatterThe science of chemistry developed from the observations on the nature and behavior of different kinds of matter, which we refer as the properties of matter. The properties we refer to in this lesson are all macroscopic properties – those that can be observed in bulk matter. On the other hand, at the microscopic level, matter is characterized by its structure – the spatial arrangement of the individual atoms in a molecular unit or an extended solid. The study of matter starts with the study of its properties A. Physical PropertiesPhysical properties are those which can be measured or observed without observable changes in the composition or identity of the substance. Now we distinguish that matter commonly exists as a solid, a liquid, or a gas and that it has mass and volume. In what other ways can you describe matter? Suppose you are asked to characterize the solids in front of you, how are you going to describe them? You probably will begin by describing their shape,size, color or degree of hardness. The characteristics that you observe without changing the matter are called physical properties. Categories of Physical Properties1. Extensive PropertiesThese properties do depend on the amount of matter present. Some of the most common types are:
2. Intensive PropertiesIntensive properties are very important, because every possible kind of matter possesses a unique set of intensive properties that distinguishes it from every other kind of matter. It serves to characterize matter. The properties given below do not depend on the amount of the matter present.
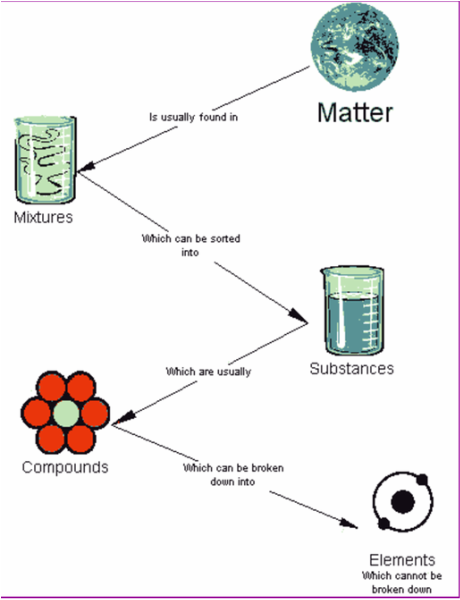
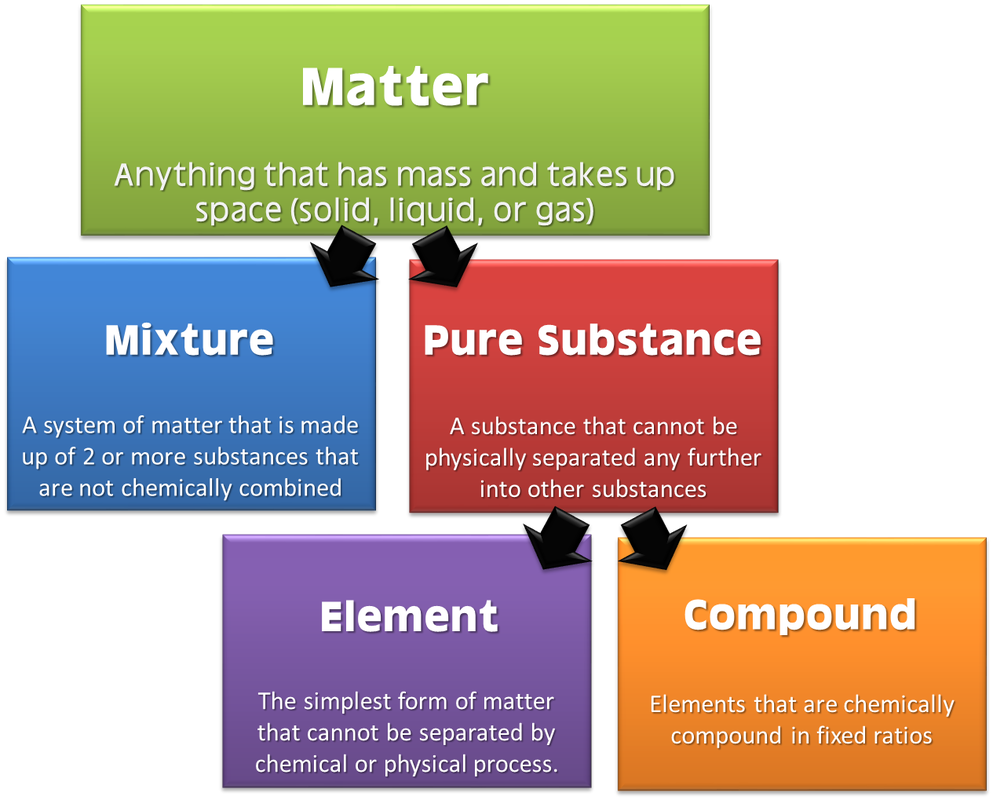
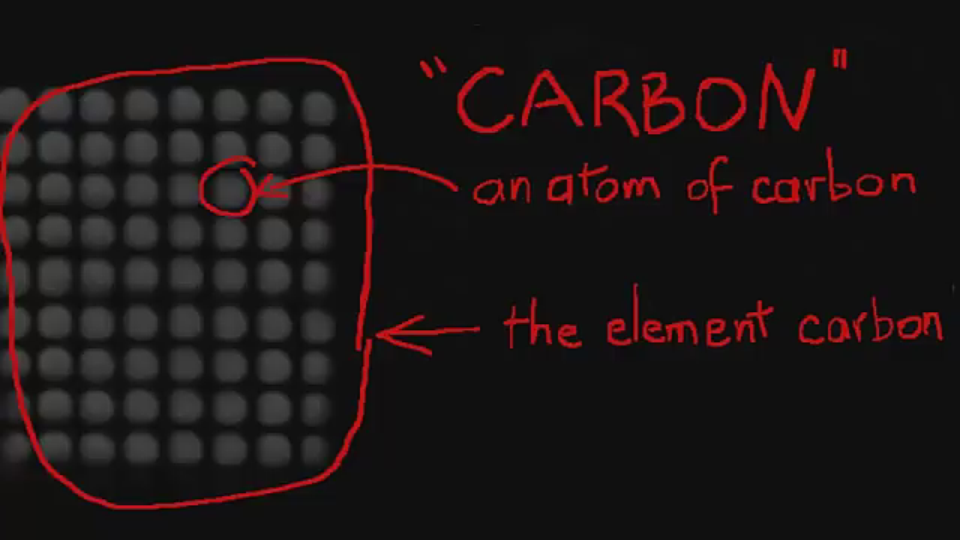
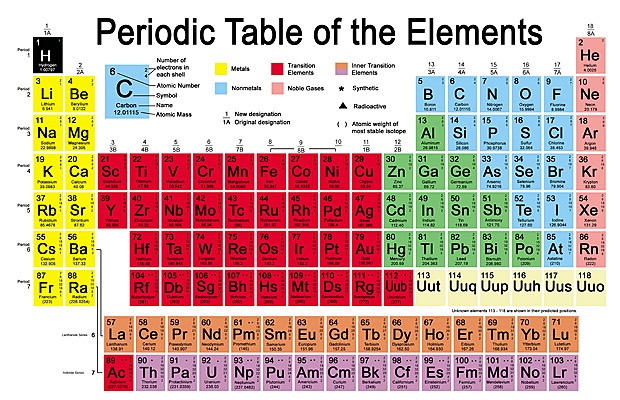
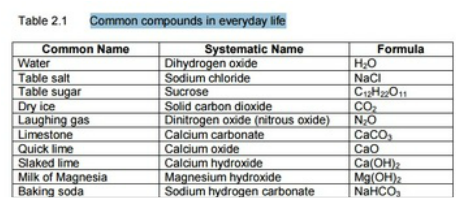
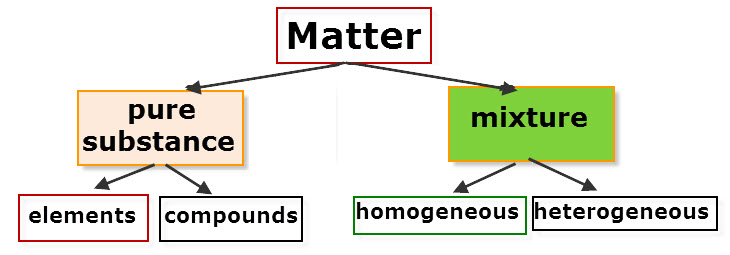
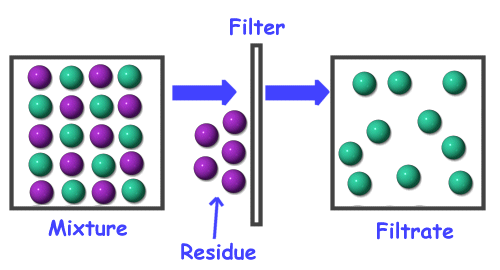
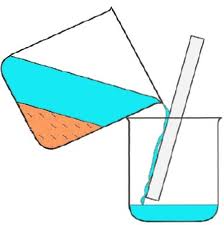
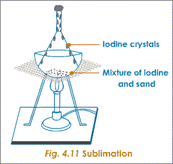
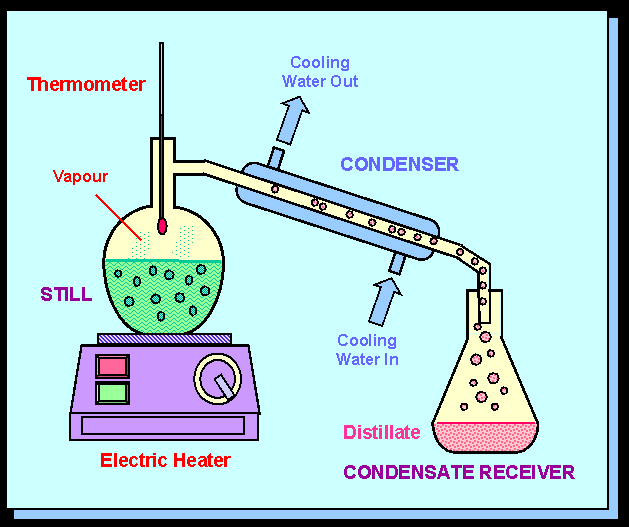
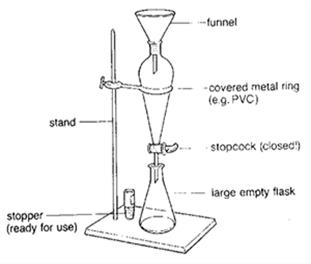
B. Chemical Properties Chemical properties require chemical change to take place. In this case, the substance changes in the identity as well as its composition. You learned that in your chemistry class how matter changes. Chemical properties of matter depend on how one substance reacts with other substances. Paper burns because it reacts with oxygen in the air. Kinds of MatterThe two general kinds of matter are pure substances and mixtures. Pure substances are further subdivided into elements and compounds while mixtures are subdivided into homogeneous and heterogeneous mixture. A. Pure substanceA pure substance refers to a sample of matter that has a distinct set of properties that are common to all other samples of that substance. A good example would be sodium chloride or ordinary salt. Once the samples of this substancehave been purified,they possess the same unique set of properties nomatter what its source (from a mine, evaporated from seawater, or made in the laboratory). 1. ElementsElements are pure substances which are composed of atoms that can not be broken down into simpler substances. They can be classified as metals, nonmetals and metalloids. The names and symbol of these elements are listed in the Periodic Table. Examples of some metallic elements are sodium, magnesium, potassium, barium and aluminum. Among the nonmetallic elements are carbon, oxygen, sulfur, chlorine and nitrogen. Among the metalloids are silicon, arsenic, boron and antimony. 2. CompoundsCompounds is a kind of pure substance which is made up of two or more elements that combined chemically and the components can be separated by chemical means but not by any physical means. The table 2.1 below shows the names of compounds that we encounter in every day life. B. MixturesA mixture, in contrast, is composed of two or more substances that can exhibit a wide range of properties depending on the relative amounts of the components present in the mixture. The components of a mixture can be easily separated by physical means because they are not chemically combined. 1.Homogeneous matter (from the Greek homo = same) can be thought of as being uniform and continuous. This is a kind of mixture in which the components form only one phase. Several homogeneous mixtures are commonly referred to as solutions. Examples of homogeneous mixtures are sugar mixed with water, milk, and etc. 2. Heterogeneous matter (hetero = different) implies non-uniformity and discontinuity. Heterogeneous mixture is a kind of mixture which forms two or more phases that can be easily seen through the naked eye. Examples of heterogeneous mixtures are sand mixed with iron fillings, air (mixture of different gasses), vinegar with oil, and etc. Methods to Separate Mixture Components1. FiltrationThis is a separation of solid components from liquid components of a mixture by using filter paper. The liquid that passed through the filter paper is called the filtrate while the solid part that is left in the filter paper is called the residue. 2. Decantation3. EvaporationThis is a separation of volatile component from the nonvolatile component by heating the mixture until the volatile evaporates. 4. Sublimation5. Distillationis a separation of the components of liquid mixture with different boiling points. It involves 2 stages and both are physical state changes a. The liquid mixture is boiled to vaporize the most volatile component in the mixture (liquid ==> gas). b. Using cold water, the vapor is cooled in the condenser to condense (gas ==> liquid) it back to a liquid (the distillate) which is collected. Distillation process can be used to purify water because the dissolved solids have a much higher boiling point and will not evaporate with the steam. Miscellaneous Separation MethodsThere are apparatus which is being used to separate mixtures and these are: 1.Separatory funnel Distillation was described in the previous discussion. Let us recall that it is used to separate miscible liquids that dissolve in each other. On the other hand, If two liquids do not mix, they form two separate layers and referred to us immiscible liquids (e.g. oil/water). This is illustrated in the diagram on the right, where the lower liquid is denser than the upper layer of the liquid. The two immiscible liquids can be separated using a separatory funnel. To separate the two liquids, the mixture is put in the separatory funnel with the stopper on and the tap closed and the layers left to settle out. The stopper is removed, and the tap is opened so that you can carefully run the lower layer off first into a beaker or any container. This leaves behind the upper layer liquid. 2. Magnet This can be used to separate sulfur from a mixture with iron. It is used in recycling to pick up iron and steel from domestic waster. Matter and its Changes Matter on Earth normally exists as a solid, a liquid, or a gas. It can be changed from one phase to another. For example, water is a liquid and it will become a solid if it is frozen. Keep in mind that as substances cool they lose heat thus, they lose energy. Ice has less heat energy than liquid water. When water is heated, it can become a gas in the form of vapor. As water warm up, they gain heat. Some materials can be changed from one form to another. For instance, when a material melts, it changes from a solid to a liquid. Matter around us undergoes a method of breaking down or synthesis. Bodies of water such as seas and oceans evaporate. Iron nails rust. Dead animals decay and shortly become part of the soil. Most industrial products and medicine these days are prepared through chemical changes. Raw materials are altered into valuable products. These alteration is accompanied by changes in materials either physical or chemical means. Look at your surrounding, almost everything around us is a by-product of changes and these changes affect our lives in many ways. A. Physical changeYou are asked to tear a piece of paper into small pieces, what happened to the paper? Is there a new material or new substance formed after tearing? How about the size and shape of the paper? Are they still the equal? The answer to these questions would give you what kind of change has taken place in the paper and this is physical change. A physical change is a change in the state of matter without any change in the substance's chemical identity, thus no new substance formed. In a physical change, there is an alteration of physical properties of an object. Physical properties include: color, odor, shape, size, texture, volume, mass, weight, and density. In the previous discussions, tearing of paper changes in its shape and size. There is no new substance formed because you can still distinguish its physical property. Another example is the water in plastic container that can be frozen when cooled and then can be returned to a liquid state when heated. When you boiled the water in the kettle, the liquid water evaporates. Now, what are the physical changes you observed?
When you boiled the water, the liquid water evaporates and turned into gas. The gaseous state is in the form of water vapor. Indicators of Physical Change
B. Chemical ChangeMatter undergoes chemical change when the composition of the substances changes either these substances combine together or breaks up. A chemical reaction is takes place in a chemical change where a new substance is formed and energy is either given off (exothermic) or absorbed (endothermic). Recall in the previous discussion about physical change on the cutting of piece of paper. We have found out that is an example of physical change because there is no new substance formed. Now, get another piece of paper, this time burn the paper. What happened after? Can you distinguish the characteristic of the paper? In this case, the burnt paper lost its original characteristics. The new substance was brittle, dark and turned into ash. The chemical composition is different from the composition of the original substance. What you observed is a chemical change. Indicators of Chemical ChangeThe following can indicate that a chemical change took place.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories
All
|









 RSS Feed
RSS Feed
