The Atomic TheoryThe Greek Philosopher Democritus (460 – 162 B.C.) expressed the belief that matter composed of very small, indivisible particle called atomos (Greek word for uncuttable). However, his idea was not accepted by many of the contemporaries particularly Plato and Aristotle. 2000 years later, the atomic concept of matter was reintroduced. John Dalton(6 September 1766 – 27 July 1844) was an English chemist, mathematician and philosopher. In 1808, he formulated a precise definition of atoms. He is best known for his initiating work in the development of modern atomic theory. The “Dalton’s atomic theory” marks the beginning of present-day era of chemistry. Five Major Points of Dalton's Atomic Theory
Dalton’s atomic theory supports the important law in chemistry and these are: 1. Law of definite proportions
2. Law of multiple proportions
3. Law of conservation of mass
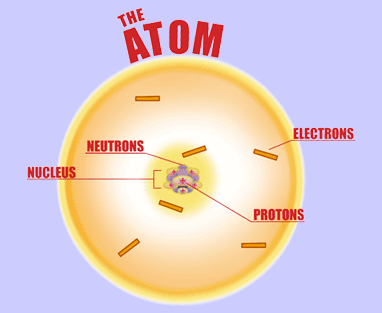
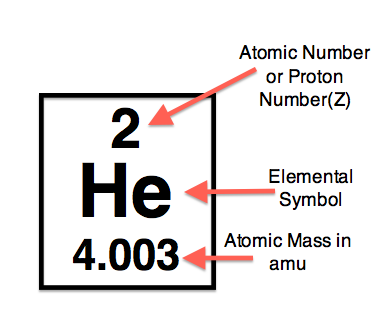
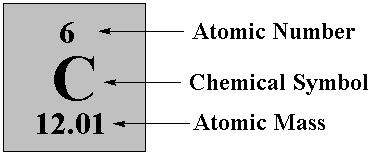
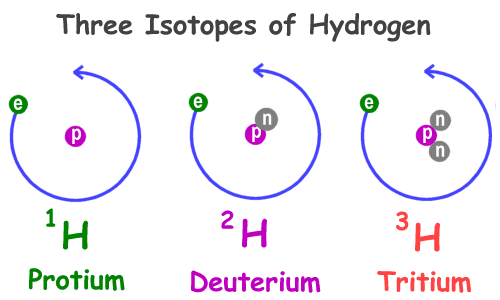
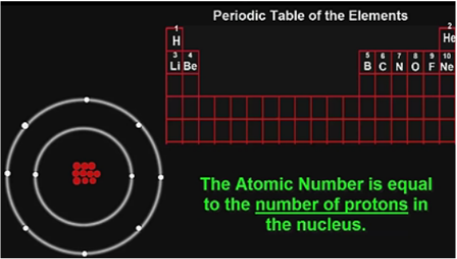
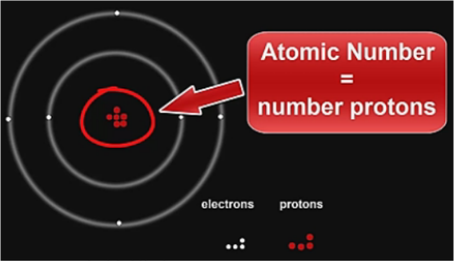
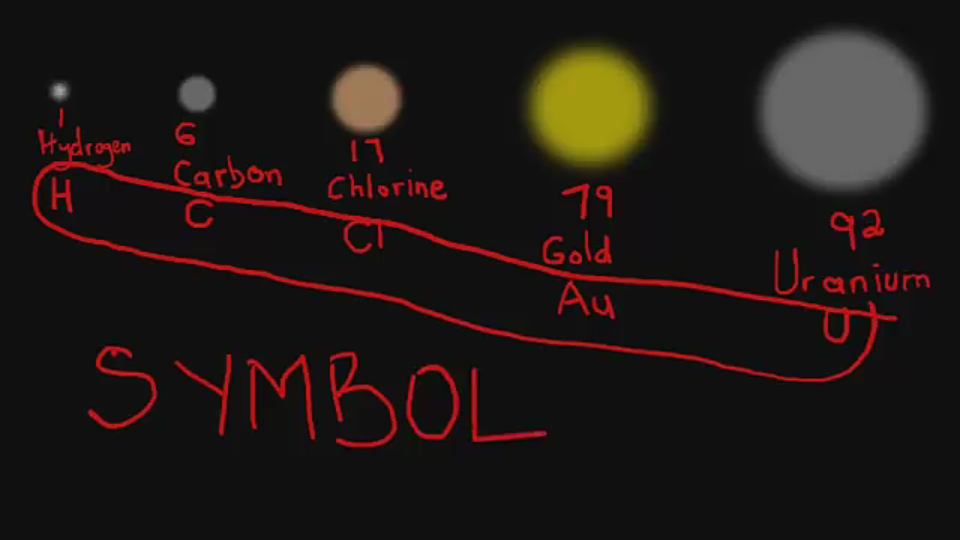
The AtomThe atom is a basic unit of matter. As mentioned earlier, It came form a Greek word “atomos” which means uncuttable, or indivisible, something that cannot be divided further. It is composed of a dense, nucleus located at the center and surrounded by a cloud of negatively charge electrons. The atomic nucleus consists of a positively charged protons and electrically neutral neutrons. A group of atoms can remain bound to each other and it will form a molecule. An electrically neutral atom contains an equal number of protons and electrons and it becomes an ion if it has a positive or negative charge. An atom is classified according to the number of protons and neutrons in its nucleus. Structure of an atomIn the early 1900s, the remarkably full acceptance of the atom came with the discovery that the atom was not indivisible after all. It is clearly demonstrated that atoms actually possess parts that gave an internal structure called subatomic particles. These three subatomic particles that atom is made up of are electrons, protons and neutrons. 1. ElectronsElectrons have a negative charge. They are smaller and more abundant than the bigger protons and neutrons. This was discovered by J.J. Thomson through his cathode ray tube experiment. 2. ProtonsErnest Rutherford together with his students made a series of experiments and investigates the nature of atoms in 1911. In their experiment, they used a thin solid foil which is surrounded by a fluorescent screen that glows when a particle touches it. The set-up was bombarded with positively charged particles. They have found out that most of alpha particles passed through the gold foil. Based on their findings, atom is made up of spaces because the alpha particles passed through. The spaces are the location where the electrons are situated surrounding the nucleus while the solid part that was hit is the nucleus where the protons are positioned. Hence, protons are positive charge and are located in the nucleus of an atom, along with the neutrons. 3. Neutrons are neutral hence no charge. Now, you might ask how did they discover neutrons if they do not have a charge. This question can be answered because of the additional weight in the nucleus of an atom. They also keep the protons in equilibrium because they hold them together inside the nucleus. Atomic number, Mass number, and IsotopesThe first thing you will need to do is find some information about your element using the Periodic Table of Elements.The atomic number is the number located in the upper left corner and the atomic weight is the number located on the bottom, as in this example for Helium: Atomic numberIt is the number of protons in an atom of an element. In our example,Helium's atomic number is 2. This tells us that an atom of krypton has 2 protons in its nucleus. The interesting thing here is that every atom of carbon contains 2 protons. If an atom doesn't have 2 protons, it can't be an atom of carbon. Adding or removing protons from the nucleus of an atom creates a different element. For example, removing one proton from an atom of carbon creates an atom of boron. On the other hand, electrons are arranged around atoms in a special way. Whatever the number of protons, it is the same as the number of electrons, thus, the electron number of Helium is also 2. IonIon is formed when an atom gain or lose electrons. It is nothing more than an electrically charged atom. An element does not change if electrons are added or remove from an atom, just its net charge. Atomic weightIt is basically a measurement of the total number of particles in a nucleus of an atom. The atomic weight is in fact a weighted average of all of the naturally occurring isotopes of an element relative to the mass of carbon-12. Mass numberLook at your periodic table of elements, unfortunately, the mass number is not listed in it. How are you going to find the mass number? To find it, all you need to do is round the atomic weight to the nearest whole number. In our example, Carbon’s mass number is 12 since its atomic weight, 12.01, round off to 12. So now you know how to find the mass number. Remember that the mass number is the number of particles in an atom's nucleus. The nucleus is made up of protons and neutrons. So, we can write: Mass Number = (Number of Protons) + (Number of Neutrons) For Carbon, this equation becomes: 12 = (Number of Protons) + (Number of Neutrons) If we only knew how many protons Carbon has, we could figure out how many neutrons it has. Wait a minute... We do know how many protons krypton has! We did that back in Step 2! The atomic number (6) is the number of protons in Carbon. Putting this into the equation, we get: 12 = 6 + (Number of Neutrons) What number added to 6 makes 12? Hopefully, you said 6. That is the number of neutrons in an atom of Carbon. The interesting thing here is that it does not create a different element by adding or removing neutrons from an atom. Rather, it makes a heavier or lighter description of that element. These different descriptions are called isotopes and most elements are actually a mixture of different isotopes. To sum up...For any given element: Atomic Number = Number of Protons Number of Protons = Number of Electrons = Atomic Number Number of Neutrons = Mass Number – Atomic Number IsotopesIsotope is defined as atoms of the same element possessing different numbers of neutrons or atoms of the same element having the same atomic number but different mass number. For example, the most common isotope of hydrogen has no neutrons at all; deuterium is an isotope of hydrogen with one neutron, and another,tritium, with two neutrons. If you want to refer to a definite isotope, you write it like this: AXZ. Where X is the chemical symbol for the element, Z is the atomic number, and A is the mass number. Ordinary hydrogen is written 1H1, deuterium is 2H1, and tritium is3H1. How many isotopes do you think can one element have? Can you give other example of an isotope? Light elements tend to have about as many neutrons as protons while heavy elements apparently need more neutrons than protons in order to attach together. Atoms with a few too many neutrons, or not quite enough, can sometimes exist for a while, but they're unstable meaning the atoms are radioactive. Radioactive isotopes undergo decay process and their nuclei change by emitting radiation in the form of particles or electromagnetic waves. IN SUMMARY
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories
All
|









 RSS Feed
RSS Feed
